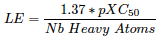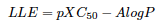Compound 1141
Identifiers
- Canonical SMILES:
CN[C@@H](C)C(=O)N[C@@H](C1CCCCC1)C(=O)N1CCC[C@H]1c1nc2c(Cc3ccccc3)cccc2s1
- IUPAC name:
(2S)-N-[(1S)-2-[(2R)-2-(4-benzyl-1,3-benzothiazol-2-yl)pyrrolidin-1-yl]-1-cyclohexyl-2-oxoethyl]-2-(methylamino)propanamide
- InChi:
InChI=1S/C30H38N4O2S/c1-20(31-2)28(35)32-27(22-13-7-4-8-14-22)30(36)34-18-10-16-24(34)29-33-26-23(15-9-17-25(26)37-29)19-21-11-5-3-6-12-21/h3,5-6,9,11-12,15,17,20,22,24,27,31H,4,7-8,10,13-14,16,18-19H2,1-2H3,(H,32,35)/t20-,24-,27-/m0/s1
- InChiKey:
YVVYRJPRGCJJLO-PPNCUWOSSA-N
External links
 46883883 |
 CHEMBL1092174 |
 24679764 |
External search

|

|

|

|

|
Bibliography (1)
Pharmacological data
| Biochemical tests | Cellular tests | PK tests | Cytotoxicity tests |
|---|---|---|---|
| 1 | 0 | 0 | 0 |
Targets
| PPI family | Best activity | Diseases | MMoA |
|---|---|---|---|
| XIAP / Smac | 6.82 | cancer | Inhibition |
Physicochemical filters
| Descriptor | Lipinski's RO5 | Veber | Pfizer's 3/75 | |
|---|---|---|---|---|
| Compliance | ||||
| MW | 518.27 g/mol | |||
| HBA | 6 | |||
| HBD | 2 | |||
| HBA + HBD | 8 | |||
| AlogP | 5.36 | |||
| TPSA | 74.33 | |||
| RB | 8 |
Radar chart
PCA : iPPI-DB chemical space
PCA : Correlation circle
Efficiencies: iPPI-DB biplot LE versus LLE
Summary
| Bibliographic ressources | Biochemical tests | Cellular tests | PK tests | Cytotoxicity tests |
|---|---|---|---|---|
| 1 | 1 | 0 | 0 | 0 |
| Ta | Structure | Name | Drugbank ID |
|---|---|---|---|
| 0.6062 | LCL-161 | DB12085 | |
| 0.5945 | Dolastatin 10 | DB12730 | |
| 0.5570 | (2S,4S,5R)-1-(4-TERT-BUTYLBENZOYL)-2-ISOBUTYL-5-(1,3-THIAZOL-2-YL)PYRROLIDINE-2,4-DICARBOXYLIC ACID | DB07199 | |
| 0.5566 | RWJ-51084 | DB03251 | |
| 0.5351 | RWJ-56423 | DB02812 | |
| 0.5024 | 1-[(4S)-4-amino-5-(1,3-benzothiazol-2-yl)-5-oxopentyl]guanidine | DB08624 | |
| 0.4585 | (2R,3R)-N^1^-[(1S)-2,2-DIMETHYL-1-(METHYLCARBAMOYL)PROPYL]-N^4^-HYDROXY-2-(2-METHYLPROPYL)-3-{[(1,3-THIAZOL-2-YLCARBONYL)AMINO]METHYL}BUTANEDIAMIDE | DB08733 | |
| 0.4488 | 4-METHYL-PENTANOIC ACID {1-[4-GUANIDINO-1-(THIAZOLE-2-CARBONYL)-BUTYLCARBAMOYL]-2-METHYL-PROPYL}-AMIDE | DB07299 | |
| 0.4483 | N-((1R,2S)-2-(5-CHLORO-1H-INDOLE-2-CARBOXAMIDO)CYCLOHEXYL)-5-METHYL-4,5,6,7-TETRAHYDROTHIAZOLO[5,4-C]PYRIDINE-2-CARBOXAMIDE | DB07629 | |
| 0.4483 | N-((1R,2R)-2-(5-CHLORO-1H-INDOLE-2-CARBOXAMIDO)CYCLOHEXYL)-5-METHYL-4,5,6,7-TETRAHYDROTHIAZOLO[5,4-C]PYRIDINE-2-CARBOXAMIDE | DB07630 | |
| 0.4440 | Lidorestat | DB07063 | |
| 0.4418 | IDD552 | DB02834 | |
| 0.4328 | (S)-2-CHLORO-N-(1-(2-(2-HYDROXYETHYLAMINO)-2-OXOETHYL)-2-OXO-1,2,3,4-TETRAHYDROQUINOLIN-3-YL)-6H-THIENO[2,3-B]PYRROLE-5-CARBOXAMIDE | DB07792 | |
| 0.4319 | 2-CHLORO-N-[(3R)-2-OXO-1,2,3,4-TETRAHYDROQUINOLIN-3-YL]-6H-THIENO[2,3-B]PYRROLE-5-CARBOXAMIDE | DB07066 | |
| 0.4274 | Oprozomib | DB11991 |